sur Jaguar Health, Inc. (NASDAQ:JAGX)
Jaguar Health Completes Crofelemer Trial for CID in Dogs
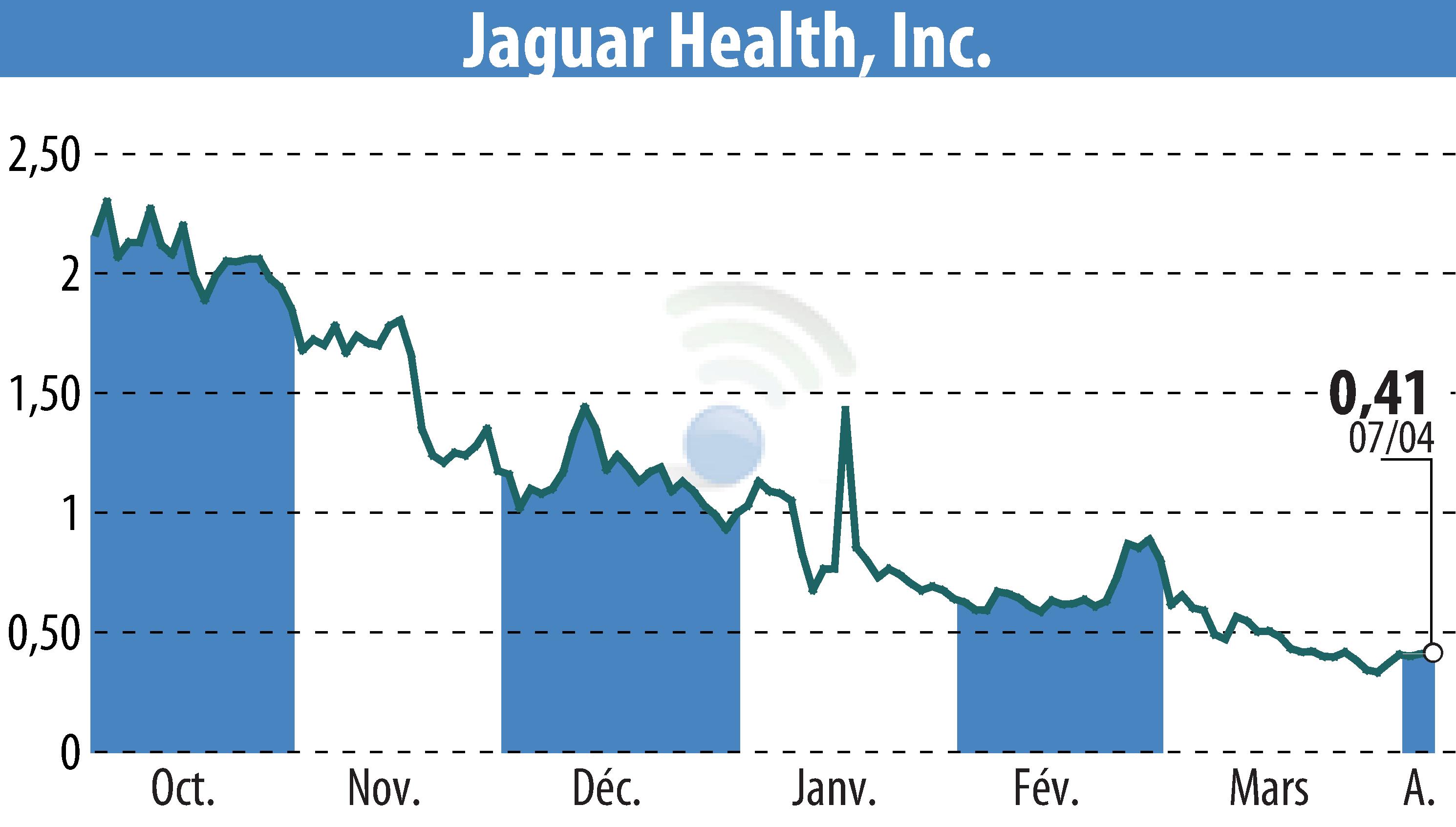
Jaguar Health, Inc. has completed its effectiveness trial of crofelemer delayed-release tablets for treating chemotherapy-induced diarrhea (CID) in dogs. Crofelemer, marketed as Canalevia-CA1, is conditionally approved for this use. The conditional approval expires on December 21, 2026.
The recent study enrolled dogs at veterinary oncology clinics across the U.S. Results are expected within a month and may support the approval process for the drug. Jaguar plans to submit the study results to the FDA in the second quarter of 2026.
A complementary study demonstrated crofelemer's effectiveness in reducing diarrhea severity in dogs treated with neratinib, a targeted cancer therapy. This study showed a statistically significant 30% reduction in diarrhea incidence and severity.
Crofelemer is not an antibiotic, minimizing concerns about antibiotic resistance. As more dogs undergo chemotherapy, effective management of side effects like diarrhea becomes increasingly vital.
R. E.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de Jaguar Health, Inc.
