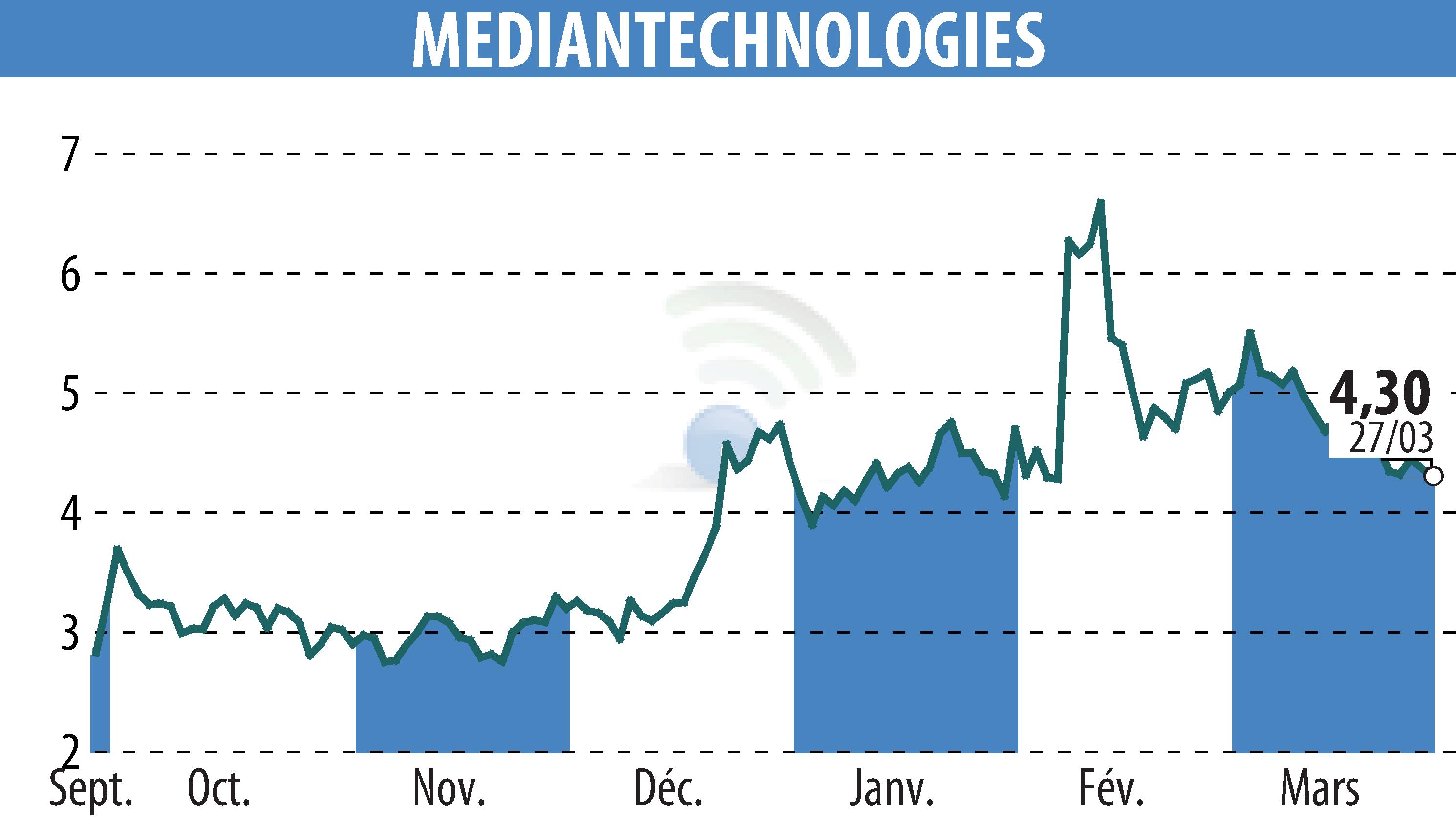
sur MEDIAN TECHNOLOGIES (EPA:ALMDT)
Median Technologies Achieves ISO 13485:2016 Certification
Median Technologies has announced it has obtained ISO 13485:2016 certification for its eyonis® medical device quality management system. This certification, issued by the French accredited body GMED, highlights the company's adherence to international regulatory standards for AI-based software as medical devices (SaMD).
The certified quality management system covers the full operational scope of the eyonis® business unit, emphasizing design, development, deployment, sales, and maintenance processes. This recognition underscores Median's capability to industrialize and scale its advanced AI-based solutions, setting a foundation for future regulatory approvals.
The certification is a critical step, following recent achievements like FDA 510(k) clearance, and reinforces the company's commitment to delivering dependable oncology solutions worldwide. It also complements previous certifications, ISO 27001:2022 and HDS 2.0, showcasing a robust system for managing sensitive data.
R. E.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de MEDIAN TECHNOLOGIES
