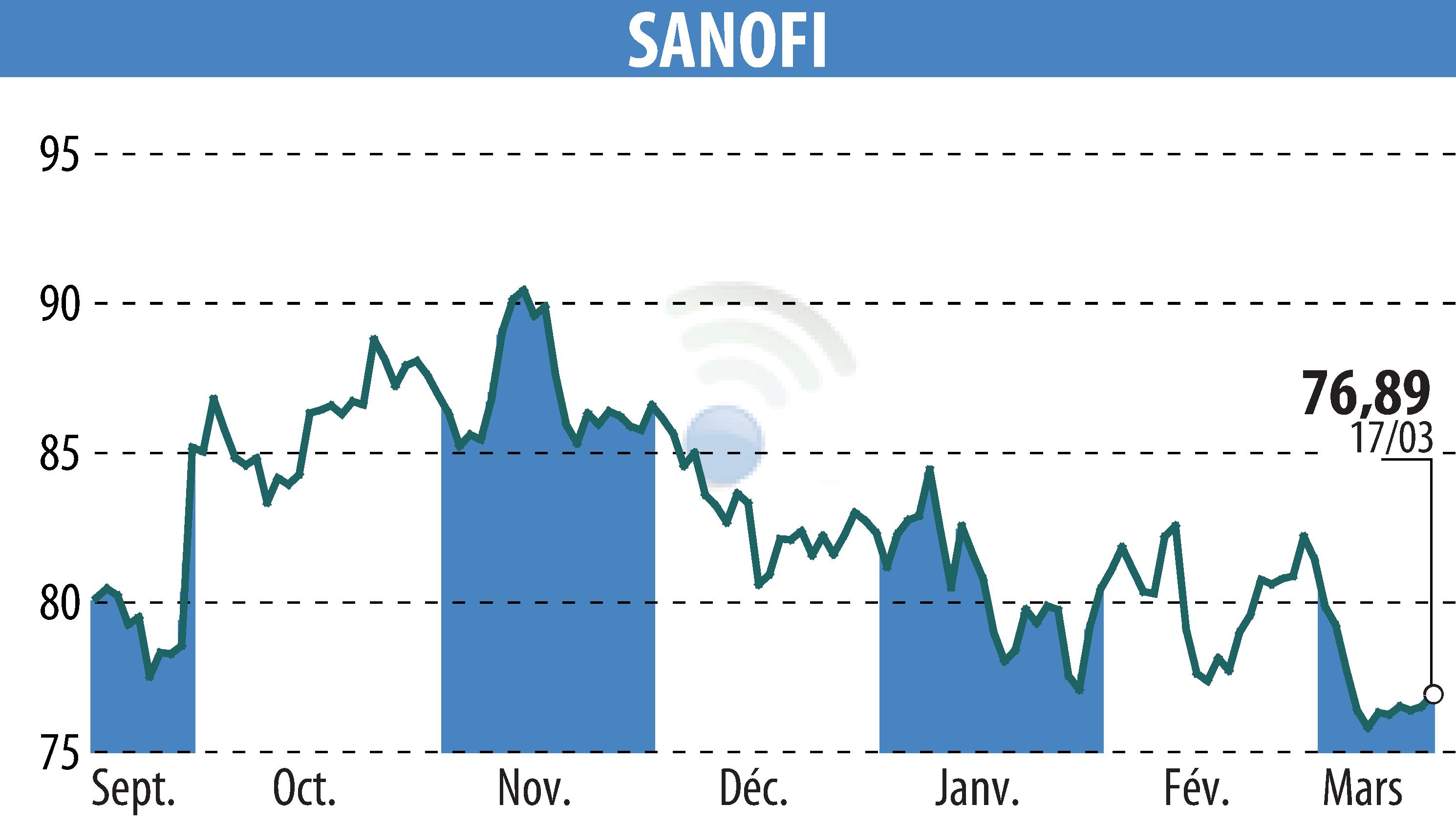
sur SANOFI-AVENTIS (EPA:SAN)
Sanofi: Breakthrough Therapy Designation for venglustat in the United States
Sanofi announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Therapy designation to venglustat, an oral treatment for type 3 Gaucher disease. This rare disease is caused by an enzyme deficiency leading to lysosomal storage, with progressive neurological symptoms. The designation is based on results from the Phase 3 LEAP2MONO study, which demonstrated an improvement in neurological symptoms in patients treated with venglustat compared to standard enzyme replacement therapy.
Sanofi emphasizes that venglustat has not revealed any new significant side effects compared to previous studies. Commonly observed adverse effects include headaches and nausea. Already designated a "fast-track" and "orphan drug," venglustat continues its development, highlighting the need for innovative solutions for neurological symptoms associated with MG3.
R. E.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de SANOFI-AVENTIS
