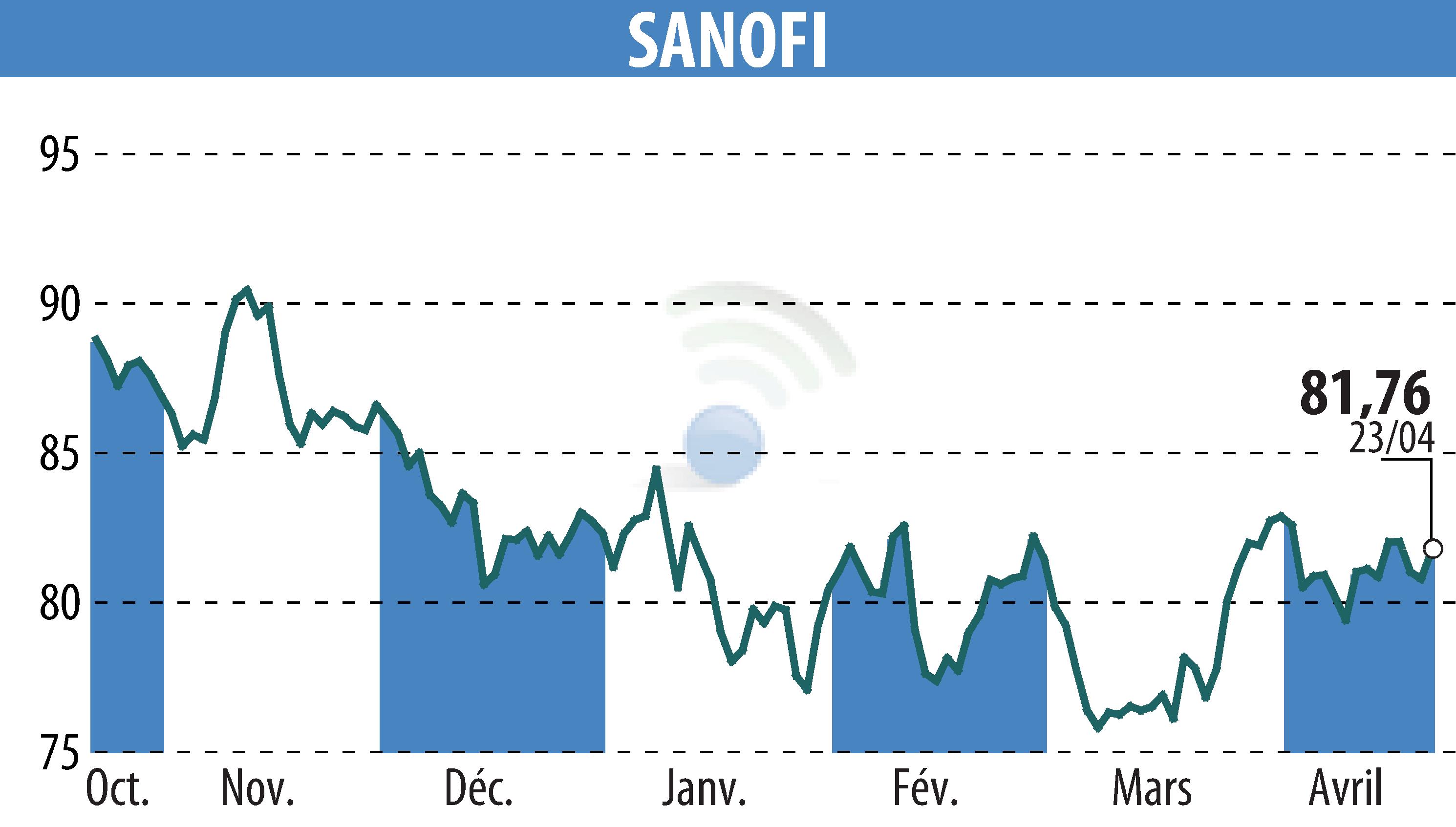
sur SANOFI-AVENTIS (EPA:SAN)
Sanofi's Cenrifki Receives Positive CHMP Opinion for SPMS
Sanofi has announced that its Bruton’s tyrosine kinase inhibitor, Cenrifki (tolebrutinib), has received a positive opinion from the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP). This recommendation concerns the treatment of secondary progressive multiple sclerosis (SPMS) without relapses. It is based on the results of the HERCULES phase 3 study, which showed a delay in disability progression.
The HERCULES study underlined the significant unmet need in managing legal disabilities affecting SPMS patients. Data from this study, along with GEMINI 1 and 2 studies focused on relapsing multiple sclerosis, supported the CHMP's recommendation.
While Cenrifki's safety profile was consistent, significant liver enzyme elevations were noted, underscoring the need for careful liver monitoring. The final decision from regulatory authorities is expected soon, with global submissions underway.
R. E.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de SANOFI-AVENTIS
