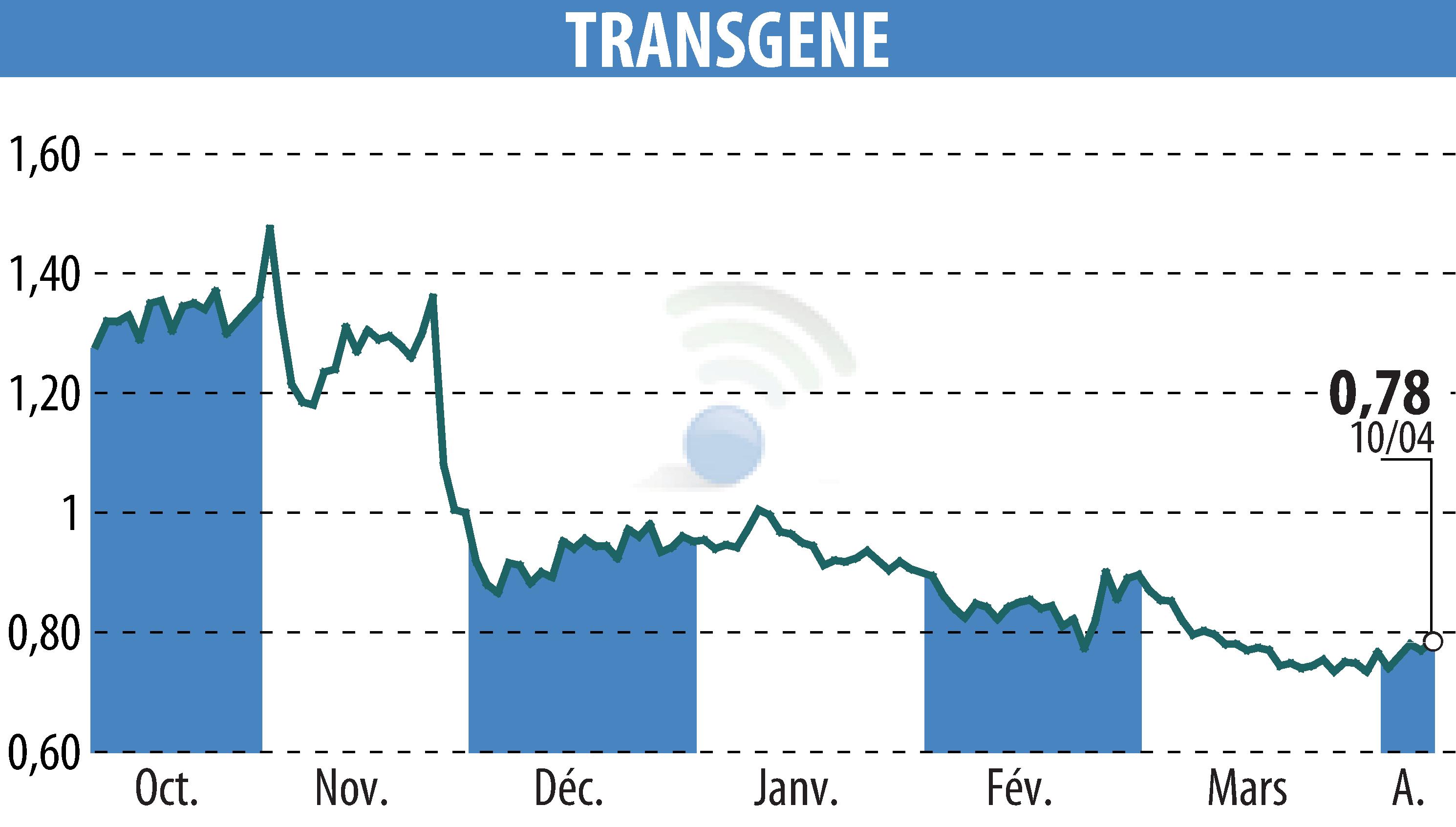
sur TRANSGENE (EPA:TNG)
Transgene Concludes Patient Randomization in Phase 2 of TG4050 Clinical Trial
Transgene, a biotech company from Strasbourg, has completed patient randomization in the Phase 2 part of its clinical trial for TG4050. This individualized neoantigen vaccine, developed from the myvac® platform, targets head and neck cancer. The trial's primary goal is assessing 2-year disease-free survival, with results expected by Q1 2028.
The trial involves 38 patients, with TG4050 designed to boost immune response post-surgery and adjuvant therapy. Previous Phase 1 results showed the vaccine was well-tolerated and maintained immune responses, with 2-year disease-free status in patients.
Dr. Alessandro Riva, CEO of Transgene, highlighted the significance of this milestone for their myvac® platform. The trial aims to demonstrate TG4050's efficacy versus watchful waiting in advanced HPV-negative head and neck cancers.
R. H.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de TRANSGENE
