sur VALNEVA (EPA:VLA)
Valneva's Lyme Disease Vaccine Shows Promise Despite Missed Trial Endpoint
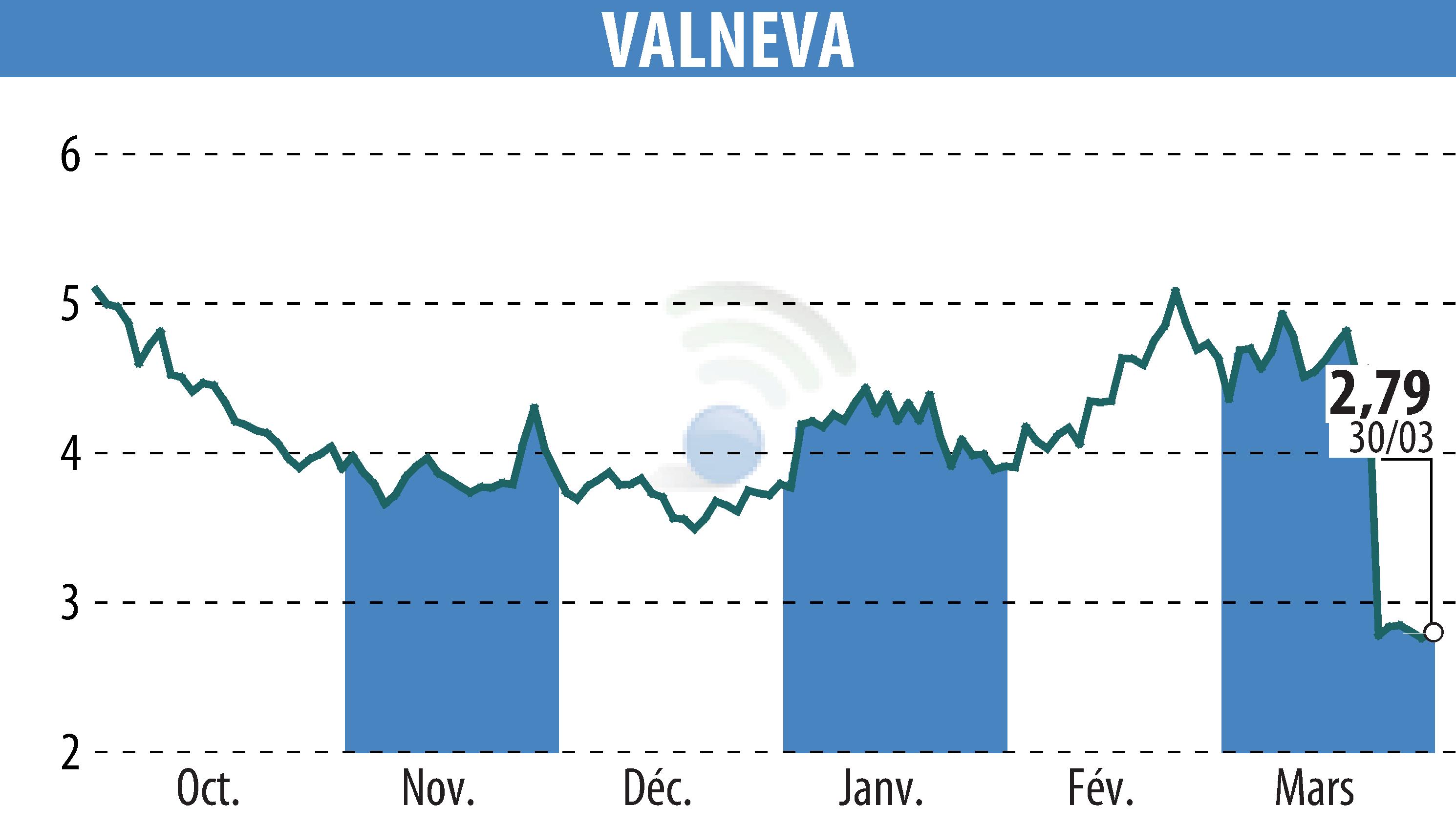
First Berlin Equity Research released an update on Valneva SE after the announcement of data from a phase 3 trial of their Lyme disease vaccine candidate, LB6V. The trial, conducted with Pfizer, showed more than 70% efficacy in individuals aged five and older and revealed no safety concerns. However, the study did not meet its primary endpoint due to a broad confidence interval of 15.8 to 93.5%. This result indicates uncertainty because fewer Lyme disease cases than anticipated occurred during the trial.
Despite missing the primary endpoint, the positive aspects include a second analysis meeting the statistical criterion, no FDA-approved alternatives, and well-tolerated results. These factors make FDA approval likely, with Pfizer planning submissions to regulatory authorities. Consequently, First Berlin Equity Research lowered the approval probability to 67% and the price target from €6.60 to €4.80, while maintaining a "Buy" recommendation for Valneva.
R. E.
Copyright © 2026 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de VALNEVA
